Distal femoral Shark bite resection of an osteosarcoma and allograft reconstruction using Patient Specific Instrumentation (PSI, Ossis Orthopaedics)
Overview
Subscribe to get full access to this operation and the extensive Bone & Soft Tissue Tumour Surgery Atlas.
Professional Guidelines Included
Learn the Distal femoral Shark bite resection of an osteosarcoma and allograft reconstruction using Patient Specific Instrumentation (PSI, Ossis Orthopaedics) surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Distal femoral Shark bite resection of an osteosarcoma and allograft reconstruction using Patient Specific Instrumentation (PSI, Ossis Orthopaedics) surgical procedure.
Osteosarcoma, Ewing’s sarcoma and chondrosarcoma are the three most common primary malignant sarcomas of bone. There are a number of other primary malignant bone sarcomas which do not fit neatly into one of the above three histiotypes, and these are generally known as ‘spindle cell sarcomas of bone’. They are rare and include such entities as fibrosarcoma, malignant fibrous histiocytoma and leiomyosarcoma of bone.
The most commonly affected anatomical sites are the femur, followed by the tibia, pelvis and humerus. The typical age for a spindle cell sarcoma of bone is 45 years and 25% of patients present with a pathological fracture. Clinical outcomes for patients with spindle cell sarcoma of bone have a better prognosis than osteosarcoma when matched for age with reportedly 67% five-year survival (Pakos et al. The ‘other’ bone sarcomas: Prognostic factors and outcomes of spindle cell sarcomas of bone. J Bone Joint Surg Br 2011;93-B:1271–8).
Limb-salvage surgery is a realistic expectation in approximately 80% of cases, even in the presence of a pathological fracture. In 20% of patients amputation as primary surgical treatment for their sarcoma is still required. The most common limb-salvage procedure is endoprosthetic reconstruction following en-bloc excision, which generally results in a reasonable functional outcome, allowing patients to bear weight and resume near normal function swiftly. Less common surgical reconstructions (in the UK at least) include massive allografts to replace the missing bone, which may be augmented with vascularised fibula autografts in more extensive cases.
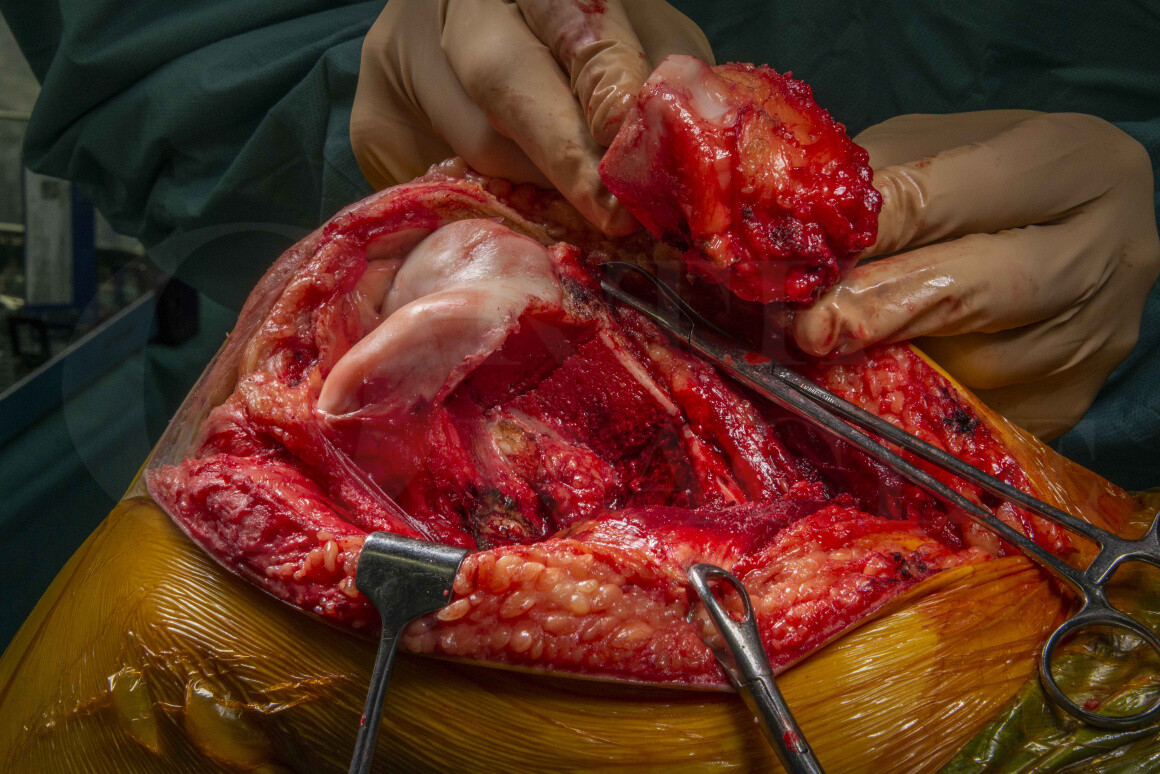
Using patient specific instrumentation (PSI) to resect and reconstruct has increased in popularity in the orthopaedic oncology world, although there are limitations to its use. However PSI does have a role for achieving complex osteotomies when performing so-called ‘shark-bite’ or hemi-cortical excisions of malignant bone tumours, where less than 50% of the meta-diaphysis is resected, especially as the allograft can be cut to perfectly match the resection.
These PSI jigs are manufactured often using 3D printers after importing the patient’s CT and MRI scans to design custom single-use jigs to perform the osteotomies, in this case from Ossis orthopaedics, but most implant companies who manufacture custom prostheses for use in orthopaedic oncology surgery will offer PSI with their implants. Limitations of these PSI jigs are surgical access as they can often be quite bulky to put into the wound; additionally the foot-print of the jig must sit with stability on the surface of the bone meaning all soft-tissues have to be cleared, which may compromise resection margins in oncology cases.
Here I describe the use of a PSI jig to perform a ‘shark-bite’ excision of a distal femoral primary bone sarcoma and massive osteo-articular allograft reconstruction, in order to preserve the native knee joint in a young adult patient.
OrthOracle readers will also find the following operative techniques of interest:
Distal femoral endoprosthesis: Stanmore Juvenile Tumour System custom implant for osteosarcoma
Proximal tibial endoprosthesis (Stryker) for osteosarcoma
Osteosarcoma of proximal humerus: Resection and epiphyseal fibula transfer
Author: Jonathan Stevenson FRCS (Tr & Orth)
Institution: The Royal Orthopaedic Hospital, Birmingham, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















