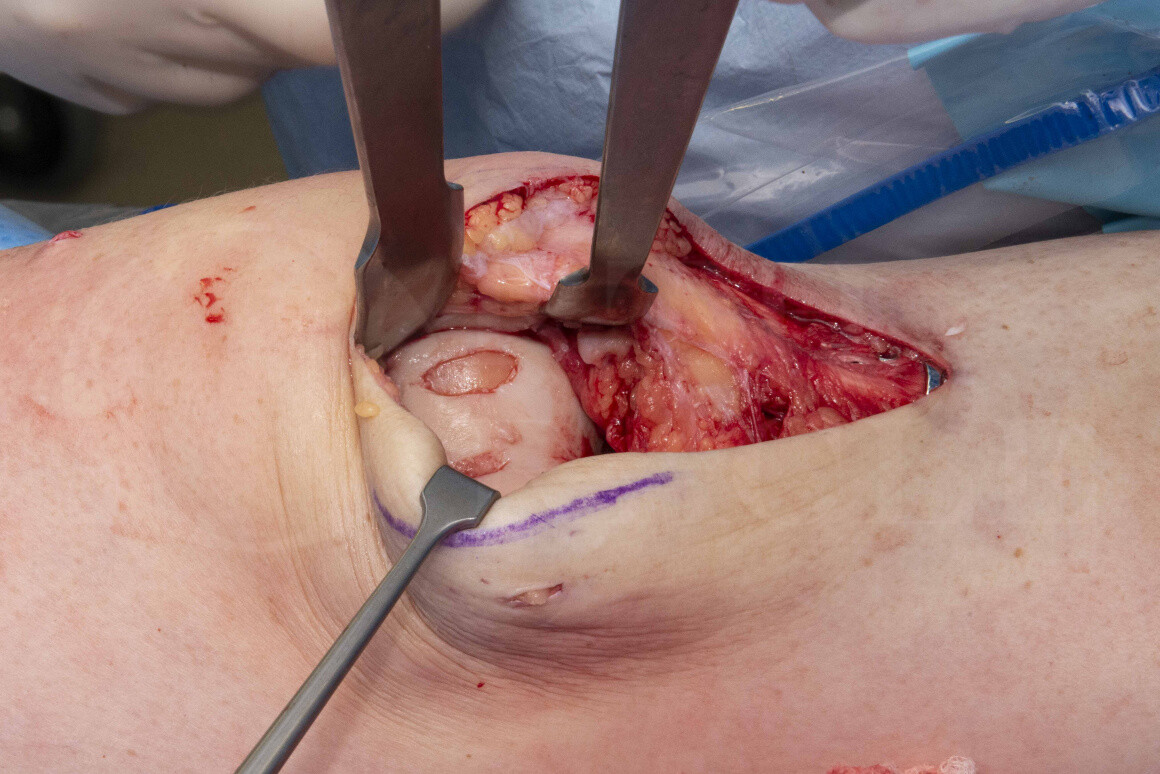
Femoral trochlea chondral lesion: Chondrogide membrane(Geistlich pharma) for chondral regeneration
Overview
Subscribe to get full access to this operation and the extensive Knee Surgery Atlas.
Learn the Femoral trochlea chondral lesion: Chondrogide membrane(Geistlich pharma) for chondral regeneration surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Femoral trochlea chondral lesion: Chondrogide membrane(Geistlich pharma) for chondral regeneration surgical procedure.
Chondral regenerative techniques have been used for many years, but due to the problems in funding for autologous chondrocyte implantation techniques, the non-cell-based techniques using membranes are gaining popularity for the treatment of focal articular defects. In distinction to focal resurfacing there is a slower recovery and some post-operative restrictions are required, but the potential of biological reconstruction without removing any native joint is appealing in the younger population. These biological reconstruction techniques usually require a period of reduced weightbearing up to 2 months and often a restricted range of movement range using a brace. The chondral membrane techniques may be used as isolated chondral surface regenerates or they may be combined with stimulation techniques which breach the subchondral bone. Similarly the chondral membrane techniques may be combined with bone grafting (autologous or allograft which is my preference) or for some surgeons synthetic bone graft substitute to reconstruct a bone void beneath a chondral lesion.
The classic patient for biological reconstruction is in the 18-50 age range with a focal chondral lesion which is symptomatic and has failed traditional non-operative treatments such as offloader bracing and injection therapies such as visco-supplementation. Steroid injection is controversial for these patients as there may be some symptomatic relief, but likely damage to the good native chondral surface and therefore this is not part of my standard practice for young chondral damage patients.
Please remember to check alignment as osteotomy is an excellent proven treatment for the malaligned overloaded joint. Chondral regenerative techniques will not work in mechanically overloaded joints without osteotomy, which is a principle this applies in the tibiofemoral as well as the patella-femoral joints which are covered on OrthOracle at Patella stabilisation : Tibial tubercle osteotomy and Medial patellofemoral ligament reconstruction.
Chondrogide is a Porcine Collagen type I/III matrix produced by Giestlich. It has been used clinically for over 15 years with a metanalysis from 2019 demonstrating its clinical efficacy in the knee with benefit maintained for at least 5 years in a population of 20-50 year old patients.
Steinwachs MR, Gille J, Volz M, Anders S, Jakob R, De Girolamo L, Volpi P, Schiavone-Panni A, Scheffler S, Reiss E, Wittmann U. Systematic Review and Meta-Analysis of the Clinical Evidence on the Use of Autologous Matrix-Induced Chondrogenesis in the Knee. Cartilage. 2019 Sep 11:1947603519870846. doi: 10.1177/1947603519870846. Epub ahead of print. PMID: 31508990.
The Chondrogide membranes are available in 3 sizes and are sold with a sterile aluminium foil template for ease of measuring the chondral defect.
Readers will also find of interest the following OrthOracle techniques:
Medial Femoral Condyle Focal Resurfacing with HemiCAP(Arthrosurface)
Knee arthroscopy and microfracture of osteochondral defect
Stem cell harvest and transplant for knee osteochondral defect (Synergy Medical technologies)
Author: James Murray FRCS(Tr & Orth)
Institution: The Avon Orthopaedic centre, Southmead Hospital, Bristol, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu



















