Revision Total Knee Replacement: Second stage using PFC / MBT with metaphyseal sleeve and stem (Depuy)
Overview
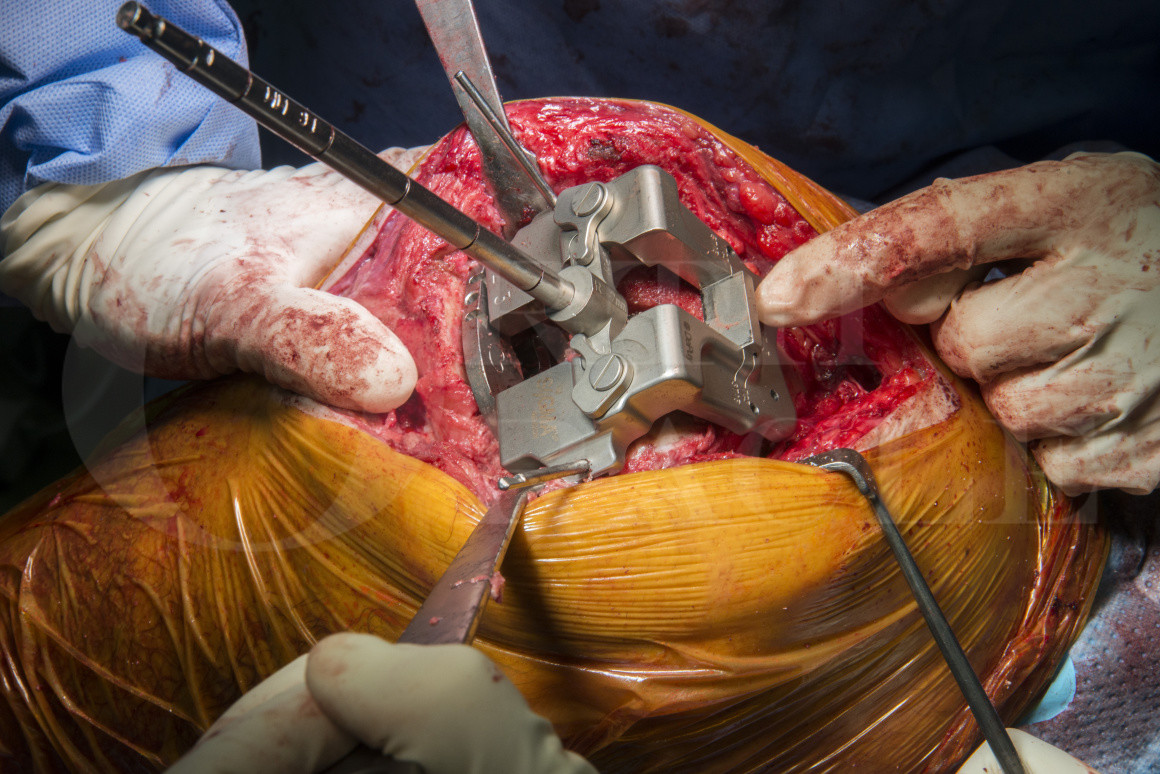
Subscribe to get full access to this operation and the extensive Knee Surgery Atlas.
Learn the Revision Total Knee Replacement: Second stage using PFC / MBT with metaphyseal sleeve and stem (Depuy) surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Revision Total Knee Replacement: Second stage using PFC / MBT with metaphyseal sleeve and stem (Depuy) surgical procedure.
This case shows the steps required to complete the second stage revision of an infected total knee replacement.
At the first stage, infected tissue was resected and a commercial antibiotic-loaded cement spacer was inserted.
This second stage takes the reader through the steps required to prepare the joint to receive the revision implant and also documents the reasoning for the revisions implants chosen and the detailed method of how to assemble and insert them.
With the increasing complexity of both infected revision surgery and the implants required to overcome the significant challenges associated with revision knee replacement, this technique goes some significant way to help the surgeon familiarise themselves with the steps required, enabling more efficient and productive surgery.
This operation was performed by Mr Richard Baker FRCS (TR & Orth) to whom I’m most grateful for the photographs.
The following report is of a mobile bearing tray (MBT), a Porocoat metaphyseal sleeve, a PFC femoral component and 2 uncemented stems, all manufactured by DePuy. I use the PFC as my primary TKA and this revision system is an extension of that family. The MBT system can articulate with either PFC or TC3 femoral components (very useful if only one component needs revising).
I also am a conservative convert to use of the metaphyseal sleeve, in limited circumstances. Metaphyseal sleeves and metaphyseal cones have developed to cope with the advent of more complex revision surgery.
In my opinion, there are two main philosophies when it comes to these void fillers and there is some overlap. Both are impacted into the bone of the tibia in an uncemented fashion. Depending on the company and the metallic properties, there is either bony ongrowth or ingrowth. Classically sleeves have been used for smaller bone voids and cones for larger defects.
A sleeve is implant specific and is attached to the tibial tray (usually via a mortise taper). A cone is used as a void filler but retains a larger central aperture, the tibial tray (and usually) a tibial stem are cemented into the metaphyseal cone. Both have their advantages and disadvantages but I personally prefer the tibial tray to be firmly attached to the tibial tray. This system is used on the MBT TKA by Depuy.
The following associated OrthOracle techniques will also be of interest to readers of this technique:
PFC Total knee replacement (De Puy-Synthes)
Revision total Knee Replacement- Legion CCK (Smith & Nephew)
Revision total Knee Replacement- Legion Rotating Hinged Knee ( Smith & Nephew)
Sleeved Total Knee Replacement for Tibial Plateau Fracture (MBT DePuy)
Author: Mr Andrew Gordon FRCS (TR & Orth)
Institution: The Northern General Hospital, Sheffield,UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















