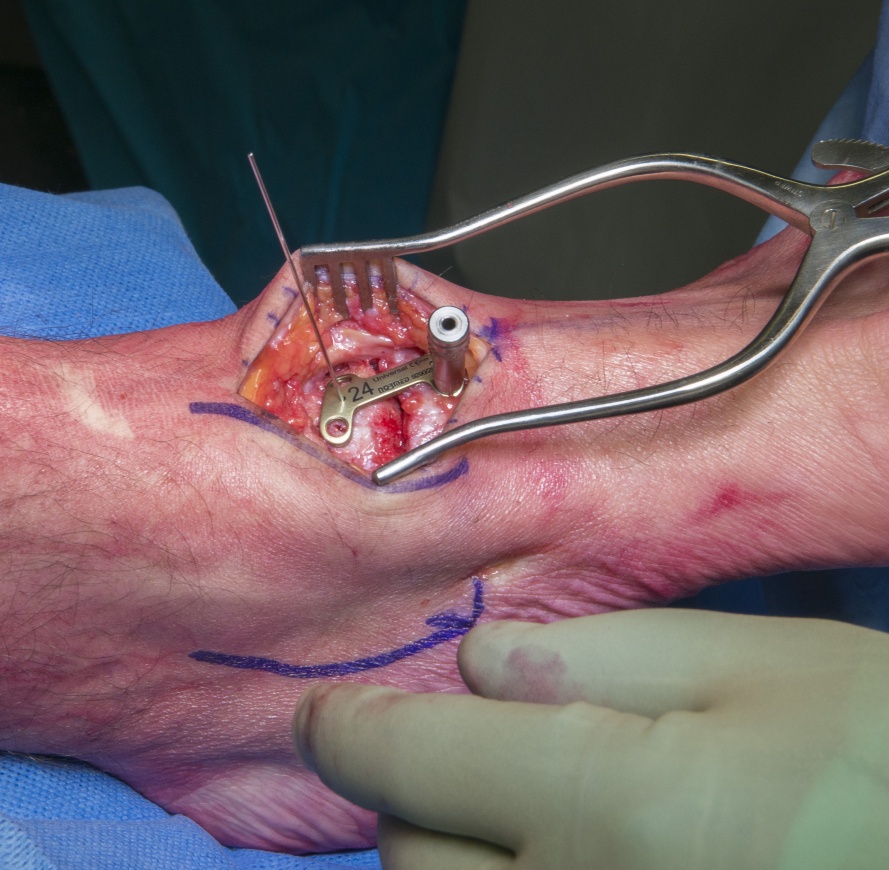
Talo-navicular fusion using Zimmer-Biomet locking plate
Overview
Subscribe to get full access to this operation and the extensive Foot Surgery Atlas.
Learn the Talo-navicular fusion using Zimmer-Biomet locking plate surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Talo-navicular fusion using Zimmer-Biomet locking plate surgical procedure.
Talonavicular osteoarthritis is a moderately common condition to be seen and treated by foot and ankle surgeons. It has many aetiologies including being the sequelae of trauma as in this case.
Painful degenerative or post-inflammatory changes at the talonavicular joint are the principle indications for talo-navicular arthrodesis. The procedure may also be used to reduce deformity in severe tibialis posterior tendonosis where a medialising calcaneal osteotomy and tendon transfer are not considered robust enough, for example in an older person with poor collagen or in those with severe deformity but without subtalar arthrosis. Union rates and clinical outcomes reported are good for isolated talonavicular fusions (see ref Diezi et al below)
I not infrequently will take iliac crest bone graft to augment my fusion. This is because this joint tolerates bone defects poorly because of the poor blood supply to both the talus and the navicular bones. In this case I prepared the crest for harvest but did not need to use it. I prefer iliac crest bone graft to that from the calcaneus or the proximal tibia in light of its greater osteoblast and precursor cell count though I am aware that it has a higher reported complication rate (see reference Chiodo et al. below)
Non operative treatments involve the use of accommodative semi-rigid orthoses, activity modification and analgesia. When this conservative approach fails operative intervention should be considered.
Chiodo CP, Hahne J and Wilson MG. Histological Differences in Iliac and Tibial Bone Graft, Foot and Ankle International May1, 2010.
Author: Mr Chris Blundell FRCS (Tr & Orth).
Institution: The Northern General Hospital, Sheffield, UK.
Author: Author: Mr Chris Blundell FRCS (Tr & Orth).
Institution: Institution: The Northern General Hospital, Sheffield, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















