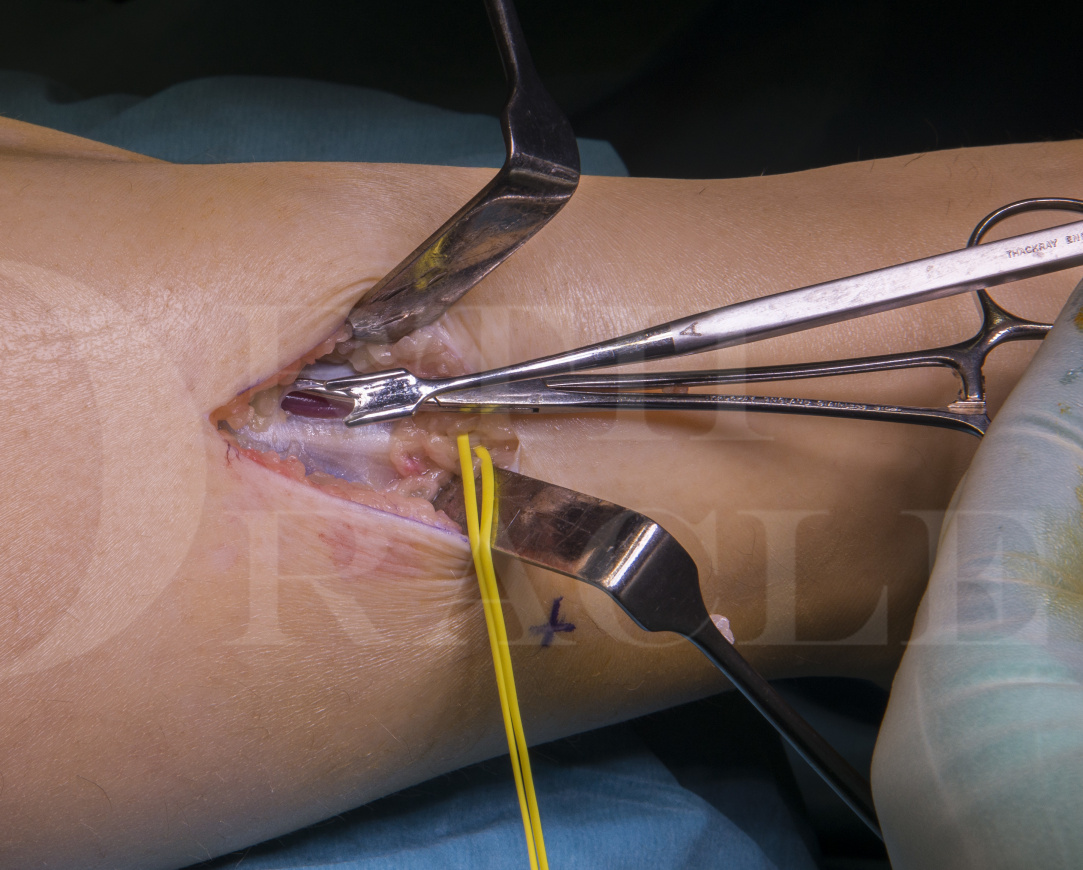
Fasciotomies of the calf for exertional compartment syndrome
Overview
Subscribe to get full access to this operation and the extensive Foot Surgery Atlas.
Learn the Fasciotomies of the calf for exertional compartment syndrome surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Fasciotomies of the calf for exertional compartment syndrome surgical procedure.
Chronic exertional compartments syndrome (CECS) of the lower limb is a condition that is generally under-diagnosed and presents with characteristic symptoms in at risk populations, such as athletes and military recruits.
CECS is characterised by exertional leg pain, which may be variably associated with pain, swelling, loss of function and distal cutaneous numbness related to specific muscle muscle compartments. The most commonly affected compartment is the anterior compartment of the lower leg(51%) followed by the lateral compartment(33%), the deep posterior compartment(13%) and superficial posterior compartment(3%).
Signs and symptoms tend to be localised to the involved muscle compartments. Presenting symptoms include cramping, aching, a feeling of muscles tightness, numbness and tingling, foot drop (often reported as slapping of the foot), and occasionally local swelling or bulging as a result of muscle herniation. These symptoms often occur in a recognisable temporal pattern in a patient, after a consistent time, distance or intensity of exercise. The pain classically becomes worse as exercise progresses, and is usually sufficiently debilitating to require an individual to stop. The symptoms reduce once exercise is stopped and have often resolved within fifteen minutes of cessation. If activity is re-commenced after a brief rest, patients commonly report a reduced length of time prior to onset of further symptoms.
Author: Nicholas Cullen
Institution: Royal National Orthopaedic Hospital, Stanmore, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















