Hallux Rigidus: Rotaglide first MTP joint replacement(Implants international)
Overview
Subscribe to get full access to this operation and the extensive Foot Surgery Atlas.
Learn the Hallux Rigidus: Rotaglide first MTP joint replacement(Implants international) surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Hallux Rigidus: Rotaglide first MTP joint replacement(Implants international) surgical procedure.
Arguably there are mainly relative indications for any first metatarso-phalangeal joint replacement given the high rates of union and patient satisfaction with the traditional surgical option of first MTP (Big toe) fusion. After a successful first MTP fusion deterioration in the longer term (and in particular from the remaining inter-phalangeal joint) is also not a common occurrence at all.
The main one practically speaking is patient preference. Interphalangeal joint arthritis with intercurrent with MTP arthritis would be one relative medical indication for replacement.
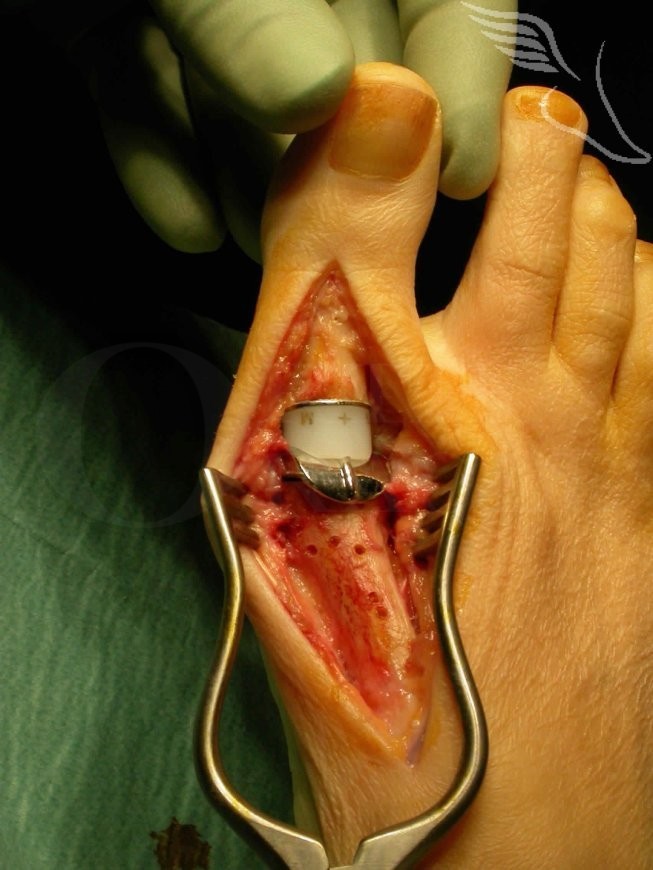
There are various designs of big toe joint replacement available ranging from hemi-arthroplasties to total 3 component joint replacements (such as the Rotaglide, Implants International). One key factor when considering which first MTP replacement to use are the published results , and longevity in particular.
A common trade-off for some element of maintained ( and also comfortable) MTP movement ( a realistic expectation after MTP replacement ) is a lower longevity with the intervention when compared to both the “gold standards” of Hip and Knee replacements and MTP fusion.
The Rotaglide MTP replacement is well jigged which assists in reproducible implantation. It is often primarily stable after implantation which allows the potential for early joint mobilisation routines in compliant patients. The approach required for the jigging is reasonably extensive.
Author: Mark Herron FRCS
Institution: The Wellington Hospital , London.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu



















