Transforaminal Interbody Lumbar Fusion (TILF) for impending cauda equina syndrome: Navigated Medtronic Solera Pedicle Screws Insertion and Globus ALTERA Interbody cage
Overview
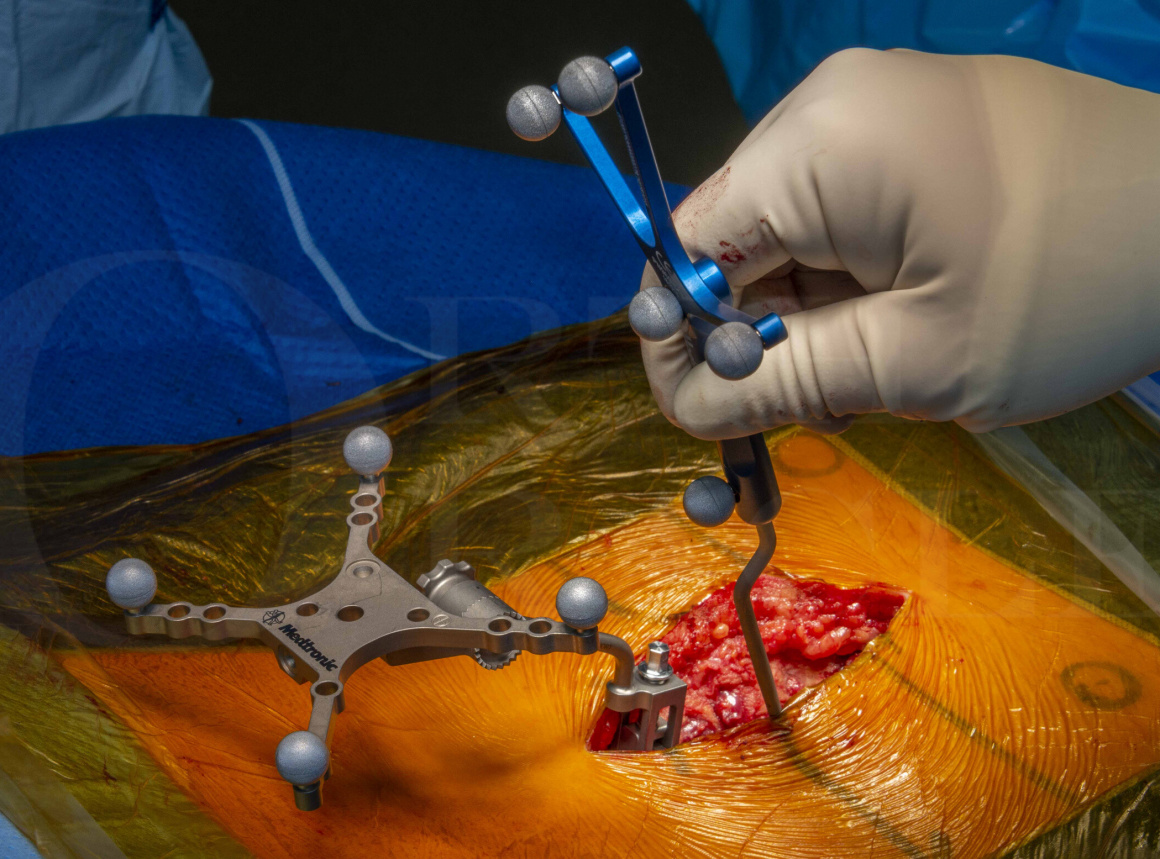
Subscribe to get full access to this operation and the extensive Spine Surgery Atlas.
Learn the Transforaminal Interbody Lumbar Fusion (TILF) for impending cauda equina syndrome: Navigated Medtronic Solera Pedicle Screws Insertion and Globus ALTERA Interbody cage surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Transforaminal Interbody Lumbar Fusion (TILF) for impending cauda equina syndrome: Navigated Medtronic Solera Pedicle Screws Insertion and Globus ALTERA Interbody cage surgical procedure.
Lumbar spinal stenosis is a very common condition but not all those affected are symptomatic. In fact, the majority are largely not affected significantly and manage symptoms by simple means. Symptoms often come on with standing or walking and improve with leaning forwards or sitting down. In some instances, patients may also experience radicular leg pain while at rest.
Spinal stenosis can occur at any level of the spine. It is most commonly caused by facet joint hypertrophy, ligamentum flavum hypertrophy, and degenerative broad disc bulges.
Spinal stenosis can be associated with spondylolisthesis, retrolisthesis, and lateral listhesis as well as any other spinal deformity which affects the space available for traversing or exiting nerve roots.
A subgroup of patients have an underlying congenital stenosis that predisposes them to becoming symptomatic if they develop mild degenerative changes later in life.
Spondylolisthesis means slipping of one spine vertebra on another. It is derived from the Greek words Spondylo, meaning vertebra, and Olisthesis (also known as the etymologically less correct listhesis), meaning slipping or sliding. The term Spondylolisthesis describes what is seen (slip of one spine vertebra on another), but not what causes it. Most spondylolisthesis are anterolisthesis (sagittal plane deformity as in this case where the slip is forward), they can however be a retrolisthesis (slip back) or lateral listhesis (slip to the side [coronal deformity]).
Surgical management options available include lumbar decompression or fusion with decompression. In some patients, a decompression procedure alone may not achieve satisfactory decompression of the nerves to improve symptoms (since in doing so the segment becomes unstable). In this situation, the surgeon needs to decide if excision of the facet joints or other bony anatomy deemed contributory to the compression will achieve desired results. If the facet joints are excised for example, instrumentation is required to achieve stability of that spinal segment.
The surgeon may also be looking to address any associated alignment/deformity problems at the time with the intention of improving the outcome of that spinal level operation but also with the hope of reducing the incidence of adjacent segment problems (jury is out if indeed this reliably occurs).
When performing a fusion operation, there are various options available to surgeons. These may involve posterior, anterior, or lateral approaches which can be performed open or via minimally invasive approaches.
In this case, given the pathology present (acute cauda equina compression at L5/S1 level) I felt a posterior approach most preferable. Due to the anatomy I felt was contributory to the symptoms I decided instrumentation of the L5/S1 level and decompression ,with insertion of the ALTERA Trans-foraminal interbody cage (TILF), would achieve my goals of surgery (those being the primary objective of adequately decompressing the cauda equina at L5/S1, improving the alignment of the L5/S1 segment and better allow for later fusion of that segment.
It will be seen later in the case that the L5/S1 disc-space was narrow. It was my opinion that an interbody cage at L5/S1 would both aid reduction of the spondylolisthesis, help indirectly decompress the L5 foramen (reduce the compression on the L5 exiting nerve roots) and improve the rate of later fusion. The ALTERA cage is expandable and therefore a smaller height can be passed into the disc space (reducing the risk of end plate damage on cage insertion since it is initially a smaller height) and once in place, expanded to a larger height. This increased height can improve the lordosis of the instrumented segment and facilitate opening of the foramen and thus allowing more space for the exiting nerve root.
ALTERA offers 8° and 15° lordotic implants and, as said, can be inserted at a minimized height (to ease insertion and navigation around the neural elements) and then expanded up to an additional 4mm height once within disc space. Of course, I would accept that 15 degrees is not truly ideal at L5/S1 and readers of the ALIF approaches will know cage lordosis of 25 degrees can be placed via that approach, but I would not generally consider ALIF cases in younger men (due to risk of retrograde ejaculation).
Readers will find the following operations of interest:
Lumbar microdiscectomy for cauda equina syndrome
L5/S1 Anterior Interbody Lumbar Fusion [Globus INDEPENDENCE MIS®]
Midline Lumbar Spine decompression (for spinal stenosis)
eXtreme Lateral Interbody Fusion (XLIF) – Nuvasive
Author: Neil Upadhyay FRCS (Tr & Orth)
Institution: The Avon Orthopaedic Hospital, Southmead, Bristol, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















