Distal tibial fracture: open reduction internal fixation with Stryker AxSOS 3 plate
Overview
Subscribe to get full access to this operation and the extensive Foot Surgery Atlas.
Learn the Distal tibial fracture: open reduction internal fixation with Stryker AxSOS 3 plate surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Distal tibial fracture: open reduction internal fixation with Stryker AxSOS 3 plate surgical procedure.
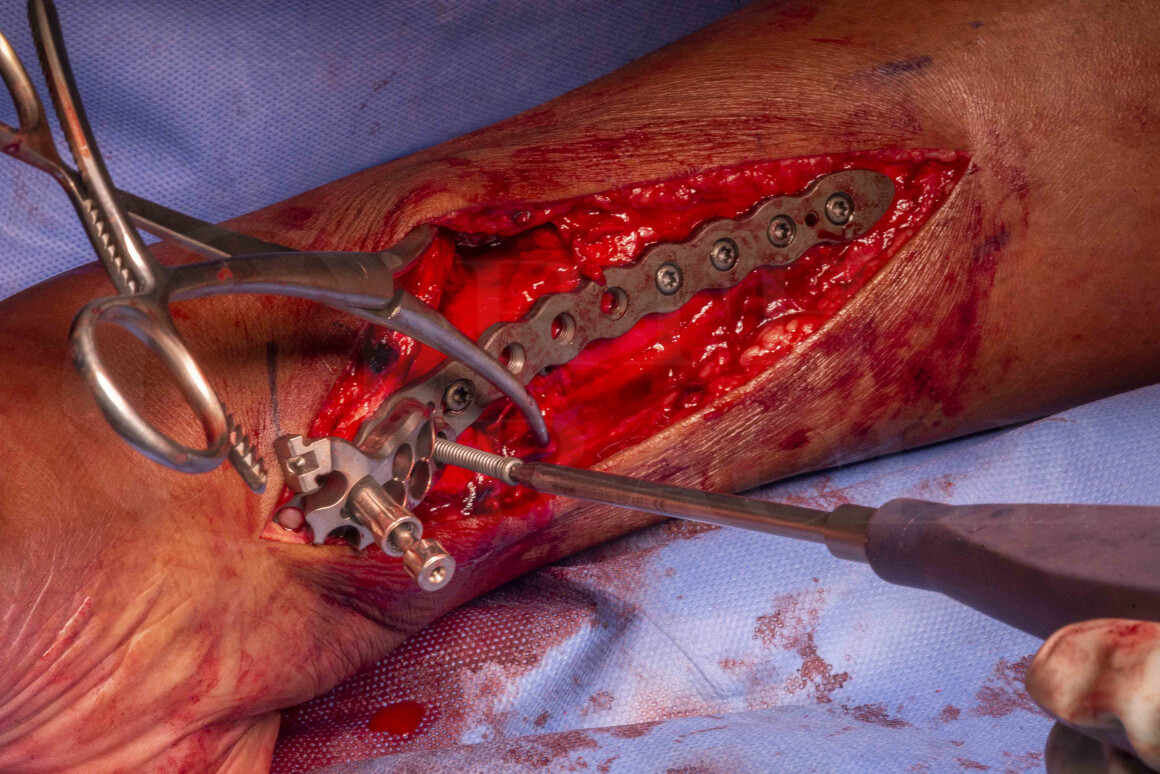
Spiral fractures of the distal tibia can pose a management challenge. Invariably, these are unstable injuries that a surgeon would struggle to manage by non-operative measures such as plaster casts or skeletal traction as achieving and maintaining a reduction is challenging. Moreover, patients do not tolerate the lengthy duration of above knee full casts or the recumbency required for skeletal traction. In addition, plain x-ray imaging reveals fracture extension into the ankle joint in about a third of all cases. Addition of CT assessment of the fracture pathoanatomy raises the appreciation of occult propagation of fracture lines into the ankle to 70%. Therefore, with these partial articular injuries (AO Foundation type 43B, see below), operative measures should be considered to achieve reduction and stability as well as restoring and preserving the articular surface.
In my opinion, I prefer internal fixation techniques that allow accurate reduction of the fracture pattern and, with the Stryker AxSOS 3 plating system, robust internal fixation using anatomically contoured plates. The Stryker distal tibial system has both medial and anterolateral plate conformations that provide surgeons with good treatment options for fixation of both extra-articular and intra-articular fractures of the tibia.
OrthOracle readers will also find the following instructional operative techniques of use:
Pilon fracture: C-type fixed using Smith and Nephew EVOS small fragment system.
Pilon Fracture: C-type fixed with Stryker AxSOS 3 Periarticular Plating System
Pilon fracture: Internal fixation using Stryker AxSOS 3Ti plate.
Ankle fracture : Fibula pro-tibia fixation technique with Stryker Variax plate.
Ankle fracture: Medial malleolar fixation with ASNIS screws
Ankle fracture: Lateral malleolar fixation using Acumed Fibula Rod System
Author: Mark Davies FRCS(Tr & Orth)
Institution: The Northern General Hospital, Sheffield, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















