Osteochondral defect (talus): Osteochondral grafting of the talus (OATS procedure)
Overview
Subscribe to get full access to this operation and the extensive Foot Surgery Atlas.
Learn the Osteochondral defect (talus): Osteochondral grafting of the talus (OATS procedure) surgical technique with step by step instructions on OrthOracle. Our e-learning platform contains high resolution images and a certified CME of the Osteochondral defect (talus): Osteochondral grafting of the talus (OATS procedure) surgical procedure.
Autologous osteochondral grafting of the Talus is normally reserved as a secondary procedure after a failed arthroscopic microfracture of the osteochondral defect.
Though there is some evidence that the high grade lesions do less well with an arthroscopic debridement and microfracture, this is a much smaller operation than open grafting and is therefore a reasonable starting point for surgical intervention.
The grafts used in this technique are cylindrical plugs “cored” out of a peripheral part of the ipsilateral knee, which can lead to secondary knee pain in some patients.
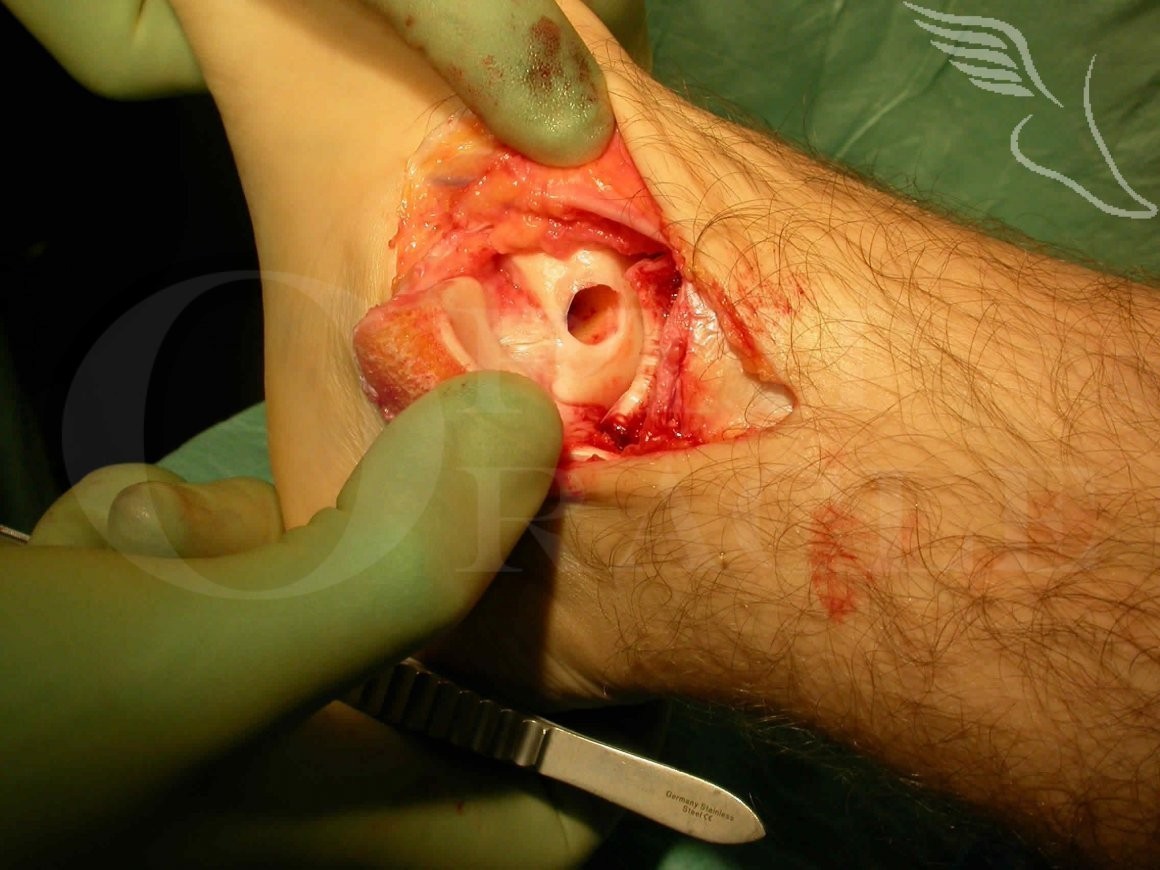
Sizing of the cylindrical grafts required(noting diameter and depth) precedes removal of the defect in this fashion, into which one or more cylindrical grafts from the knee are carefully impacted.
It is key that the Osteochondral grafts are placed as precisely (and flush) with the existing articular surface as possible, which in most cases means a malleolar osteotomy will be required to allow exact placement.
Secondary interventions are also on occasion required post grafting, such as steroid injection and on occasion further arthroscopy, and a return to normal and full function is not likely for at least 6 months post surgery.
Broadly speaking success rates of 85% or so can be anticipated, and in the longer term, as with all osteochondral surgery more minor symptoms may persist and late deterioration can occur.
OrthOracle readers will also find on interest the following associated instructional operative techniques:
Ankle arthroscopy using the Smith and Nephew Guhl non-invasive ankle distractor
Stem cell harvest and transplant for knee osteochondral defect (Synergy Medical technologies)
Knee arthroscopy and microfracture of osteochondral defect
Author: Mark Herron FRCS
Institution: The Wellington Hospital , London, UK.
Clinicians should seek clarification on whether any implant demonstrated is licensed for use in their own country.
In the USA contact: fda.gov
In the UK contact: gov.uk
In the EU contact: ema.europa.eu
Online learning is only available to subscribers.



















